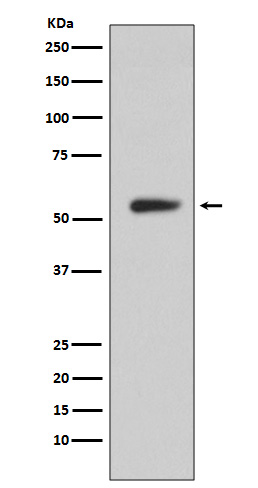
产品名称
CA9 (14N17) Rabbit Monoclonal Antibody
别名
MN; P54/58N; CAIX; CA9; Carbonic anhydrase IX; CAH9;
纯度
Affinity-chromatography
存储缓冲液
Rabbit IgG in phosphate buffered saline , pH 7.4, 150mM NaCl, 0.02% New type preservative N and 50% glycerol. Store at +4°C short term. Store at -20°C long term. Avoid freeze / thaw cycle.
Human Swissprot No.
Q16790
注意事项
CA9 Antibody is for research use only and not for use in diagnostic or therapeutic procedures.
组织表达
Expressed primarily in carcinoma cells lines. Expression is restricted to very few normal tissues and the most abundant expression is found in the epithelial cells of gastric mucosa
细胞定位
Nucleus. Nucleus, nucleolus. Cell membrane; Single-pass type I membrane protein. Cell projection, microvillus membrane; Single-pass type I membrane protein. Note=Found on the surface microvilli and in the nucleus, particularly in nucleolus
功能
Reversible hydration of carbon dioxide. Participates in pH regulation. May be involved in the control of cell proliferation and transformation. Appears to be a novel specific biomarker for a cervical neoplasia.